What Is Corrective Action?
Corrective action is an aspect of quality management that aims to rectify a task, process, product, or person’s behavior when they produce errors or deviate from an intended plan. They can respond to individual actions or to entire projects.
Essentially, corrective actions can be thought of as improvements to an organization to eliminate undesirable effects. For example, in HR for higher education, corrective action also applies to individual employees and functions to communicate to the individual what aspects of attendance, unacceptable behavior, or performance require improvement.
Corrective action along with preventive action, or CAPA, are concepts within Good Manufacturing Practice (GMP) and Hazard Analysis and Critical Control Points/Hazard Analysis and Risk-Based Preventive Controls (HACCP/HARPC), approaches commonly associated with pharmaceuticals, medical devices, and food safety. CAPA is also used to ensure compliance with FDA 21 CFR 820.100 and other regulations. Documenting problems and taking corrective actions continue to be a requirement of ISO 9000 and related standards, such as AS9100 (the aerospace standard).
The corrective action process includes clear identification of a problem and thorough documentation of the resources and steps required to mitigate the immediate symptoms. Its primary goal, though, is to find and solve the root cause of the problem. Ideally, corrective action addresses only critical problems and recurring issues, significant supply concerns, problems that endanger health or safety, or circumstances in which a customer requests a change for form, fit, and function.
- Benefits: Although the process may seem bureaucratic and drawn out, a formal corrective action offers benefits:
- It walks you through the process, so there’s no need to reinvent the problem-solving wheel.
- The corrective action document helps detail steps for solving a particular problem.
- The corrective benefits process adds transparency to the activity and empowers teams.
- It captures experience and changes for future events and development.
- Drawbacks: If implemented poorly, corrective action becomes a bureaucratic exercise in which corrective action requests are sometimes filled out for minor incidents. Corrective action also runs the risk of focusing on symptoms rather than root causes. In addition, a team may perceive it as a non-vital activity to be conducted by one or two team members during their ever-elusive “free time.”
How Does Corrective Action Contrast with Preventive Action?
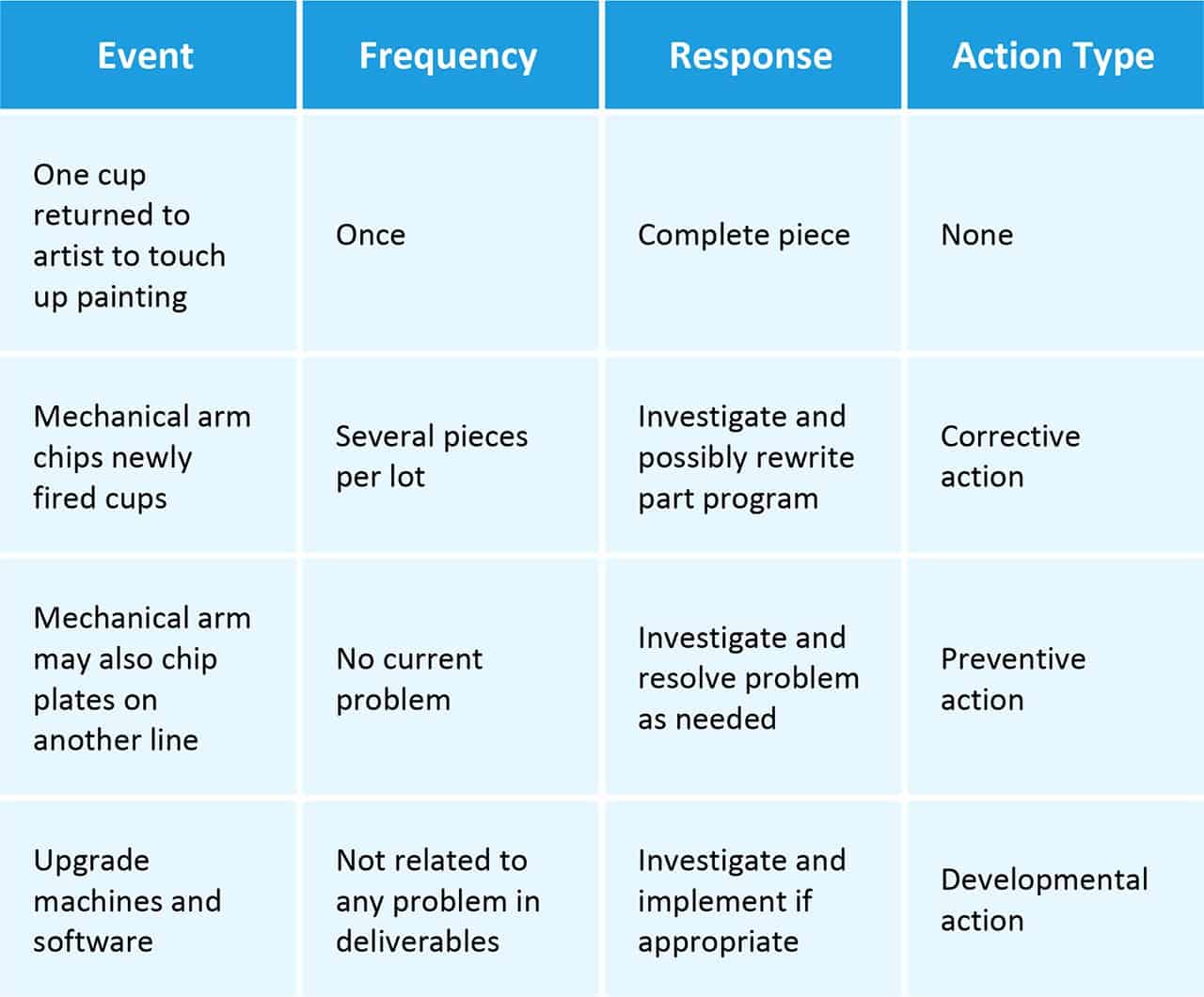
Corrective actions fix an existing problem or nonconformance, whereas preventative actions are proactive steps taken to prevent potential problems. Corrective actions occur in response to an error while preventative actions may occur before one.
-
Developmental Actions: We may view developmental actions as preventive in nature, although their principle aim is process or product improvement. As for corrective actions, the aim of the effort should be well defined, and progress and outcomes should be documented. Good documentation for new projects can help communicate the purpose to the team and capture lessons learned for future efforts.
-
Recommended Actions vs. Required Actions: In some cases, we may specify corrective actions as either recommended or required. Recommended actions can be reviewed and then accepted, rejected, or postponed. Required actions usually stem from a regulation or an internal guideline or policy. You must implement required actions — if you cannot, you must provide written justification.
What Is the Meaning of Corrective Measures?
Corrective measures are a response to problems that result in high levels of nonconformance. Corrective and preventive measures are often conflated, but preventive measures focus on avoiding issues by promoting a compliant culture and encouraging best practices.
What Is the Difference Between a Correction and a Corrective Action?
A correction is an immediate response to a known problem. A corrective action is a more measured response to an undesirable situation meant to address the cause and prevent the recurrence of said situation.
What Is Involved in Corrective Action?
Corrective action is commonly used when employees fail to meet performance standards. These actions can include warnings, suspension, demotion to a lower classification, and dismissal. Often, the process is combined with coaching as part of escalating consequences.
What Is Corrective Action in the Workplace?
Corrective action has many applications in the workplace and may be applied in different ways depending on the department. The following are examples of how you use corrective action in certain situations.
- HR Application: In HR, managers often apply corrective action after they have attempted counselling and coaching to modify an employee’s behavior (including behavior that is well outside the norm of what can be expected). In HR and in white collar establishments, pros tend to view corrective action as a disciplinary process. However, other departments, such as production in manufacturing, may take the opposite approach and desire to encourage the appropriate use of corrective actions in processes as part of continual improvement.
- Healthcare Application: When used in healthcare delivery situations, an audit by a government regulatory agency may instigate corrective actions. Some feel that a good corrective action plan can not only prevent audits and sanctions, but even legal action. The Centers for Medicare and Medicaid Services (CMS) specify a corrective action process to regulate payment errors and Children’s Health Insurance Program (CHIP) errors.
- EPA: A particular implementation of corrective actions includes remediation through the Environmental Protection Agency by way of the Resource Conservation and Recovery Act (RCRA). Specific examples include a process for adding wastewater collection to building sites and a procedure for processing soil remediation for toxic sites.
- Manufacturing for Medical Products: Medical devices and pharmaceuticals are governed by Food and Drug Administration (FDA) regulations. As such, CAPA is used to remove and avoid deficiencies in products.
What Is a Corrective Action Request?
A corrective action request is a formal request to cease the cause of a nonconformity in an item, process, or service. A detailed corrective action request can include different levels of concern based on the nature of the issue.
A CAR can originate with a customer complaint, an audit or inspection, or a production line event. Often, such as when a manufacturer deals with a supplier, the sourcing agency first discusses the problem before issuing the request. Requests can also include multiple levels of noncompliance. For example, dog nail clippers with a grip that is not comfortable for humans may not be as critical as clippers that pinch the dog’s toe pads, although they can both be issues that need correcting.
What Is a Corrective Action Plan?
A corrective action plan describes how you plan to resolve a problem or nonconformity. It details the necessary resources to correct a problem in the most cost-effective and cost-efficient way. The plan’s objectives and benefits include the following:
- It provides a standard way to address deficiencies.
- It offers premade templates that describe what types of information you need in your plan.
- It provides a process to start, research, implement, and close out a corrective action program.
- It clarifies team member or contractor responsibilities.
- It specifies what types of issues require a corrective action plan.
What Should Be Included in a Corrective Action Plan?
A corrective action plan should be very specific and include a concise statement that identifies the issue, the desirable conditions going forward, and a plan to resolve the issue.
A corrective action plan can be general: For instance, you’ll need to write more broadly when summarizing long documents, and speak in broader terms when providing detailed guidelines for addressing different severities of hazardous waste sites throughout the country and generating the paperwork required for permitting construction in such conditions. A plan or the template for dealing with troubles may detail interim measures to mitigate problems before you find a more comprehensive solution. Deadlines also apply to the creation of corrective action plans. For example, regulatory entities may impose longer lead times, whereas issues in factories may require shorter turnaround times. A corrective action plan may also include the following information:
- Stakeholders
- Resources available to solve the problem
- Constraints
- Due dates
- Metrics for completion
- Progress updates
What Is the Corrective Action Process?
The corrective action process helps you create, implement, and verify a corrective action plan. It includes steps to describe the problem, determine its causes, and implement solutions to prevent recurrence.
Andy Nichols says the concept of corrective action grew out of customer audits of suppliers. Problems, also known as nonconformities or deficiencies, would require corrective actions. The automotive manufacturing industry in particular has long provided suppliers with formats for problem solving, including the 7-step problem-solving method and the Eight Disciplines method (8D), a process developed by the Ford Motor Company in the 1980s. 8D now includes a preliminary preparation step.
The process can be described in a simpler way:
- Step 1: Describe the problem or deficiency. Explain the who, what, where, when, and why. Describe the desired outcome and how the problem can be fixed.
- Step 2: List the individuals responsible for taking corrective actions, how they report on progress and problems, to whom they report, and when they report.
- Step 3: Create measurable, achievable solutions with realistic deadlines that focus on the root cause.
- Step 4: Implement your actions and manage those actions to completion.
- Step 5: Close out the corrective action process with a debrief to determine what was done and to inform the team of changes.
- Step 6: Capture lessons learned for future problems and projects.
Using a template makes it easy to create your corrective action plan. We’ve included a free, downloadable one for your use.
Download Corrective Action Plan Template
To begin, you need to gather a team. Andy Nichols suggests that cross functional teams add particular value in bringing perspectives that are outside the specific problem area. For example, in a production line, the solution to a problem may lie in a stage before or after the actual problem.
In some cases, when dealing with certain federal regulations, such as for Medicaid and CHIP, a state may be required to create a standing review panel to check corrective action plans. In other circumstances, a large company may also choose to create its own corrective action review board. Consider all ideas as you brainstorm. A cost benefit analysis may be necessary to determine the feasibility of solutions.
When considering the team and the solution, Nichols also believes that understanding people’s problem solving styles is vital: Who needs a clean sheet of paper to begin, and who just jumps in? Sometimes a project or process should be torn down and reengineered, and sometimes time and resources don’t permit a complete reworking.
In performing a root cause analysis, it may also be helpful to understand how the problem affected the product or service before the nonconformity you identified. To determine a solution, you may also need an analysis tool, such as 5 Whys, an Ishikawa diagram (also called a fishbone diagram), or a scatter diagram. Corrective actions for regulatory compliance may even require you to perform statistical analysis to find and document problems.
Download 5 Whys Template
Excel | Word | Smartsheet
Download Ishikawa Diagram Template
The bottom line is that corrective action won’t produce a lasting resolution if it merely involves a manager filling out forms.
Best Practices for Writing Corrective Action Plans
Andy Nichols provides some thoughts on the best approach to writing corrective action plans:
- Create a Clear Problem Statement: “Very often, people focus on symptoms. That’s a human thing: ‘I’ve got a pain, I’ll take a painkiller.’ It’s very human not to find a true cause, a root cause.”
- Update All Your Documents: Often, corrective actions include changes to procedures or design, but no one remembers to update the range of pertinent documents, including the specifications. Later on, when questions arise, the team refers back to the spec for guidance, and either forgets or never knows that an update occurred.
- Let the People Brainstorm: “When the true nature of the problem has been revealed and everyone feels like they’ve contributed a huge amount to helping their work lives and the organization, they feel positively energized by being given a problem and solving it. In the quality profession, we tend to be very focused on filling out a form or the eight steps or 5 Whys or whatever, and we completely overlook the human aspect of it. Frequently, taking corrective action is focused on form filling and not on releasing the power of people’s intellects and teamwork.”
How Do You Write an Action Plan?
To write an action plan, first set your goals and identify the steps needed to achieve them. Make changes to your processes to ensure these steps can be followed, then monitor and adjust as needed.
An action plan identifies a goal and the people, resources, and activities are necessary to achieve it. By defining each resource requirement and breaking the effort into steps, a goal appears more achievable. An action plan should include the following steps:
- Set SMART goals: Goals that are achievable and measurable.
- Prioritize tasks required to meet the goals.
- Identify high-level steps needed to accomplish your goals.
- Use lists to track tasks that must be finished each day.
- Verify that you have met the goal.
- Set a deadline for completing the effort, and, if relevant, set intermediate deadlines for tasks.
You can find an assortment of action plan templates in a variety of formats here.
Improve Your Corrective Action Plans with Work Management in Smartsheet
Empower your people to go above and beyond with a flexible platform designed to match the needs of your team — and adapt as those needs change.
The Smartsheet platform makes it easy to plan, capture, manage, and report on work from anywhere, helping your team be more effective and get more done. Report on key metrics and get real-time visibility into work as it happens with roll-up reports, dashboards, and automated workflows built to keep your team connected and informed.
When teams have clarity into the work getting done, there’s no telling how much more they can accomplish in the same amount of time. Try Smartsheet for free, today.